The FDA accelerated its approval of Bruton's kinase inhibitor Jaypirca
2023-02-02
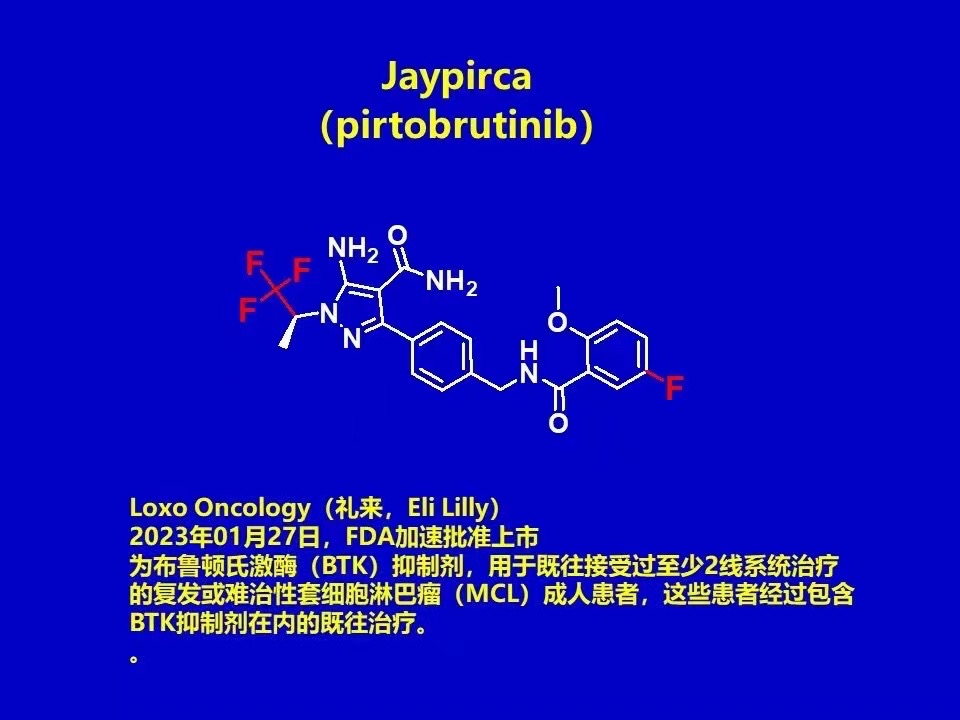
2023 The first fluorine-containing drug was approved. 2023.01.27 Eli Lilly's Loxo Oncology announced accelerated FDA approval of its BTK inhibitor Jaypirca (pirtobrutinib), For adult patients with relapsed or refractory mantle cell lymphoma (MCL) who have previously received at least 2-line system therapy after prior treatment including a BTK inhibitor (Jaypirca is the first and currently only non-covalent reversible BTK inhibitor approved by the FDA).