Vijoice is the first FDA-approved PRS treatment
2022-07-06
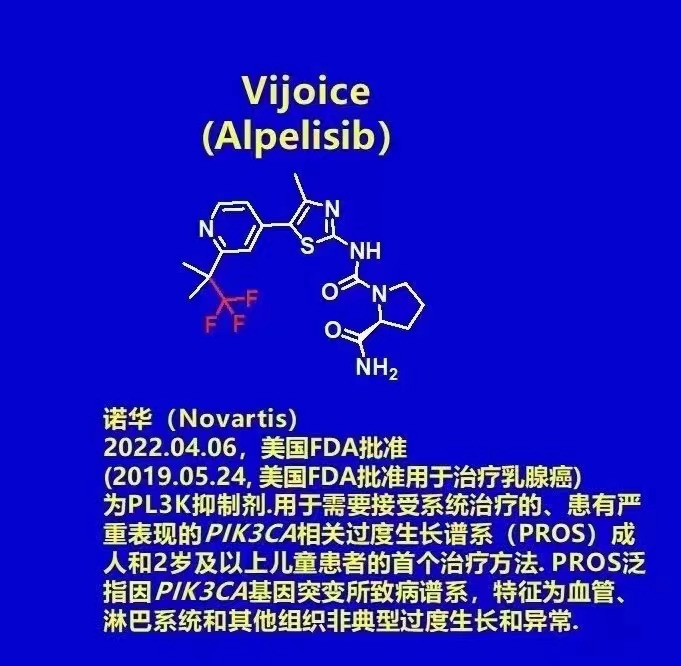
2022.04.06, Novartis announced that the U.S. Food and Drug Administration (FDA) has granted accelerated approval to Vijoice (alpelisib) for adults and children 2 years of age and older with severe manifestations of the PIK3CA-related overgrowth spectrum (PROS) requiring systemic therapy. Vijoice is The first PRS treatment drug approved by the U.S. FDA. PROS generally refers to the pathogenic spectrum caused by mutations in the PIK3CA gene, which is characterized by atypical overgrowth and abnormalities of blood vessels, lymphatic systems and other tissues. (Note: 2019.05.24, the U.S. FDA approved alpelisib for use for the treatment of breast cancer)
